|
Furthermore, their inhibition in villus cells in a rabbit model of inflammatory bowel disease (IBD) is at least in part secondary to altered Na extruding capacity of the cell due to diminished Na-K-ATPase activity. Previous studies have shown that Na-nutrient co-transport processes in the BBM, namely Na-glucose (SGLT1), Na-alanine (ATB0), Na-glutamine (B0AT1), Na-taurocholate (ASBT) and Na-adenosine (DMT1) are dependent on the BLM Na-K-ATPase for their optimal activity. Transport of different ions (H +, Ca 2+, Cl –, PO 4 2−, SO 4 2−), nutrients like glucose, amino acids and vitamins, certain nucleic acids, bile acids, and neurotransmitters across the plasma membrane are all dependent on Na-K-ATPase activity. This establishes a Na + gradient that is responsible for driving other secondary transport processes across the brush border membrane (BBM). 
Na-K-ATPase transports three Na + out of the cell in exchange for two K + into the cell, thus maintaining a high level of intracellular K + and low intracellular Na + concentration. Among the acquired transporters, Na-K-ATPase, a basolateral membrane (BLM) transporter, plays a vital role in regulating ionic homeostasis, cell volume and maintaining membrane potential. 
Nutrient, electrolyte and fluid absorption primarily occur through the villus cells while the crypt cells are thought to be primarily secretory.ĭuring the differentiation process, enterocytes acquire more transport properties, and are physiologically able to absorb more nutrients compared to undifferentiated crypt cells. The enterocytes comprise of undifferentiated crypt cells, which proliferate and differentiate to mature villus cells. While the intestinal epithelium is composed of multiple specialized cell types including goblet cells, enteroendocrine cells, Paneth cells, and enterocytes, only enterocytes are responsible for nutrient absorption from the intestinal lumen. This altered affinity is likely due to increased phosphorylation of the α1 subunit, specifically at serine and tyrosine residues.Īn essential function of the mammalian small intestine is nutrient absorption. These data indicate that as enterocytes mature from crypt-like to villus-like in culture, the functional activity of Na-K-ATPase increases secondary to altered affinity of the α1 subunit to extracellular K +, in order to accommodate the functional preference of the intestinal cell type. However, Na-K-ATPase α1 phosphorylation levels on serine and tyrosine, but not threonine, residues gradually increased. mRNA abundance and Western blot studies showed no change in the levels of Na-K-ATPase subunits α1 and β1 from 0 to 4 days post-confluent cells. Na-K-ATPase activity gradually increased as IEC-18 cells matured in vitro from day 0 (crypts) through day 4 (villus) of post-confluence. Therefore, this study aimed to determine the mechanisms involved in the functional transition of Na-K-ATPase during the maturation of crypts to villus cells. 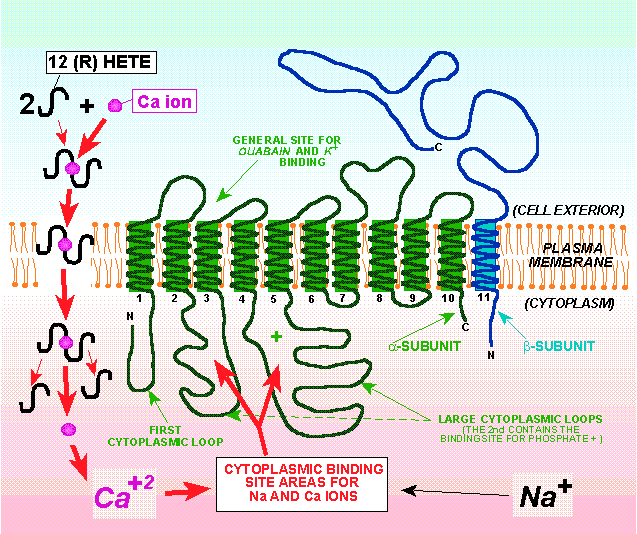
However, the mechanism of increased Na-K-ATPase activity during the maturation of enterocytes is not known. As cells mature from crypts to villus, Na-K-ATPase activity doubles, to accommodate for the increased BBM Na-dependent nutrient absorption. The cell membrane is able to conform tightly over the majority of structures with various shapes while maintaining its integrity.ģD nanostructures biointerface cell cross-section cell membrane deformation cell penetration electroporation.Na-K-ATPase on the basolateral membrane provides the favorable transcellular Na gradient for the proper functioning of Na-dependent nutrient co-transporters on the brush border membrane (BBM) of enterocytes. Here we provide a multifaceted characterization of the cellular membrane's mechanical stability when closely interacting with high-aspect-ratio 3D vertical nanostructures, providing strong evidence that vertical nanostructures spontaneously penetrate the cellular membrane to form a steady intracellular coupling only in rare cases and under specific conditions. Despite the fact that recent advancements in the fabrication of artificial biointerfaces have yielded an enhanced understanding of this interface, there remain open questions on how the cellular membrane reacts and behaves in the presence of sharp objects on the nanoscale. The dynamic interface between the cellular membrane and 3D nanostructures determines biological processes and guides the design of novel biomedical devices.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
